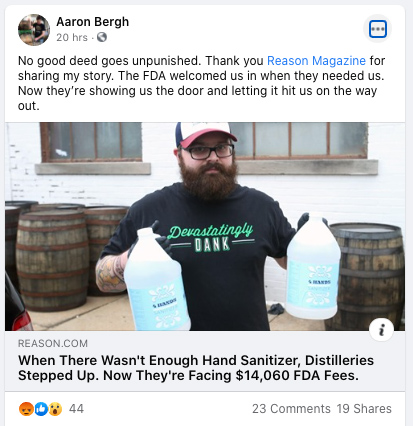
Report: Craft distilleries faced with $14,000 in FDA fees for making hand sanitizer

Aaron Bergh of Calwise Spirits / file photo
–Reason Magazine reports–
For many American craft distillers, 2020 was already one of their worst years ever. The COVID-19-related closure of tasting rooms and cocktail bars, loss of tourism, and the inability to offer in-store sampling slashed their sales revenue. Then this week, the Food and Drug Administration (FDA) had one more surprise in store: The agency delivered notice to distilleries that had produced hand sanitizer in the early days of the pandemic that they now owe an unexpected fee to the government of more than $14,000.
“I was in literal disbelief when I read it yesterday,” Aaron Bergh of Calwise Spirits in Paso Robles told Reason Magazine. “I had to confirm with my attorney this morning that it’s true.” In March, his local distillery began making hand sanitizer for hospitals, first responders, businesses, and residents.
At issue is a provision of the CARES Act that says distilleries that produced sanitizer have been classified as “over-the-counter drug monograph facilities.” The act also enacted user fees on these facilities to fund the FDA’s regulatory activities. For small distillers, that means ending the year with a surprise bill for $14,060 due on Feb. 11.
Read the full story at Reason.com
Shortly after the news stories broke about the fees, the US Department of Health and Human Services issued a response to address them. “Small businesses who stepped up to fight COVID-19 should be applauded by their government, not taxed for doing so,” said HHS Chief of Staff Brian Harrison on Twitter. “I’m pleased to announce we have directed FDA to cease enforcement of these arbitrary, surprise user fees. Happy New Year, distilleries, and cheers to you for helping keep us safe!”